Project Description
Epigenetic modeling/remodeling of cancer metastases and tumor immune contexture to improve efficacy of immunotherapy
AIRC 5xmille: Metastatic disease: the key unmet need in oncology
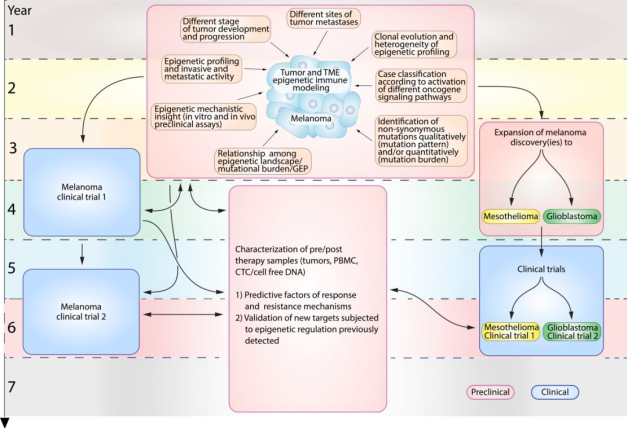
Cancer progression and metastatization is a complex process in which genetic alterations were initially demonstrated to assume a significant role; however, more recent evidence point to tumor microenvironment (TME) and host’s immune system as additional players in cancer progression. In this already multifaceted scenario, epigenetic dysregulations are most recently emerging as potential novel hallmarks of cancer metastatization. Indeed, a reciprocal interplay between tumor epigenome and genetic alterations is being suggested, and different tumor microenvironment (TME) components are being identified as signals capable to modulate cancer epigenome and immune cells trafficking at tumor sites. Epigenetic events are also increasingly recognized to down-regulate cancer immunogenicity and host’s tumor immune recognition, thus providing cancer cells with additional immune escape mechanisms ultimately contributing to their metastatization, and to therapeutic failure of immune check-point(s) blocking (ICB) drugs. These premises suggest that tumor epigenome remodeling is a feasible and promising therapeutic intervention in metastatic disease.
Basing on such a premise and on the hypothesis that pharmacological targeting of cancer cells, TME and immune system epigenome can dynamically re-shape tumor-TME cross-talk, inhibit tumor progression, remodel immune responses towards effective anti-tumor immunity, and reverse ICB resistance, the aims of this project are: i) in-depth characterization of epigenetically-regulated mechanisms regulating the functional immune-relationship” among tumor, TME, and immune cells contributing to cancer metastatization, progression, and resistance to ICB; ii) mechanistic insights to the role of altered epigenetic regulation and of specific epigenetic-modifier genes in promoting tumor progression and immunotherapy resistance; iii) developing innovative combinations of epigenetic drugs with ICB. These objectives will be pursued by conducting integrated epigenome, transcriptome, and mutational landscape analyses of melanoma, mesothelioma and glioblastoma (GBM) tissues in order to: a) establish correlations with tumor progression and metastasis, tumor immune contextures, and resistance to immunotherapy; b) identify new pharmacologically targetable pro-metastatic and immunosuppressive epigenetic processes and epigenetic modifier genes. In-vitro and in-vivo models will be used to test whether pharmacological targeting of tumor and immune system epigenome can re-shape the tumor-TME cross-talk towards effective anti-tumor immunity. Hypothesis-driven, epigenetically-based ICB combinatorial trials will be implemented in metastatic melanoma, mesothelioma and GBM.
Through this scientific approach, the project would hopefully take to define integrated profiles (epigenome/gene expression/mutational profile/immune contexture) identifying patients at differential risk of tumor progression for personalized clinical care and the selection of most appropriate candidates to current and novel ICB. One goal should be rescuing responsiveness to ICB of metastatic patients by the identification of highly-innovative, epigenetically-based “personalized” ICB regimens in melanoma, and subsequently in mesothelioma and GBM where the ICB efficacy is beginning to be explored. Indeed, identifying druggable, epigenetically-driven, immune dysregulation mechanism(s) sustaining cancer metastatization and resistance to ICB, will provide substantial practical benefits to advanced cancer patients, improving their comprehensive clinical management and providing the scientific basis for “patient-tailored” immunotherapeutic strategies.
Partners:
Dr. Maio Michele, Azienda Ospedaliera Universitaria Senese, Siena (PI); Dr. Anichini Andrea, Fondazione I.R.C.C.S. Istituto Nazionale dei Tumori, Milano; Prof. Massi Daniela, Università degli Studi di Firenze, Firenze; Prof. Palmieri Giuseppe, Consiglio Nazionale delle Ricerche, Sassari; Dr. Pfeffer Ulrich , I.R.C.C.S. Azienda Ospedaliera Universitaria “San Martino”, IST – Istituto Nazionale per la Ricerca sul Cancro, Genova; Italy.