Project Description
BIO-TAILOR (BIOliquid TAILOR study) – Clinical Trial
Dabradenib and Trametinib in circulating free DNA BRAF mutated positive melanomas: a prospective phase II study
Genomic DNA from biopsy of malignant tissues – either from the primary tumors, metastases, or cytological samples – is the standard material to determine somatic mutations and other genetic alterations before starting any targeted therapy. In addition to the tissue biopsy, which remains the gold standard for molecular diagnostic approaches, the “liquid biopsy” can represent a supplemental, or perhaps an alternative, source of DNA to identify the clinically relevant cancer mutational landscape. Indeed, this non-invasive approach may facilitate repeated monitoring of disease progression and treatment response, serving as a means to guide targeted therapies based on detected actionable mutations in patients with advanced or metastatic solid tumors. Liquid biopsies can capture the entire heterogeneity of the disease and offer additional, valuable prognostic and predictive information (Crowley, 2013; Karachaliou, 2015).
Intra-tumor heterogeneity has been recognized as a general characteristic of many cancers. From the molecular point of view, the existence of a major population of genetically different neoplastic cells and subclones has been widely demonstrated, in several models including melanoma (Lin, 2011; Colombino, 2012). The larger is the tumor size the higher is the probability of genetic-molecular heterogeneity.
Recent evidences support a model in which melanomas are in essence polyclonal, that is, include a mixture of cells that may or may not harbor the BRAF mutation, essentially demonstrating that may exist intra-tumor heterogeneity for BRAF mutations, and that changes of BRAF status over time might occur (Marieke, 2016; Santiago, 2016; Das Thakur, 2013).
Identification of patients with a BRAF mutational status shift is thus required for more accurate and effective treatment of advanced melanoma patients. In recent past years, most liquid biopsy approaches have been investigated to perform mutation analysis on circulating tumor DNA (ctDNA) samples. Among others, a study on a large collection of patients revealed that BRAF-mutated ctDNA was detected in >75% of late stage melanoma patients with BRAF mutated tumours. Moreover, absence of BRAF-mutated ctDNA at baseline seems to be associated with higher response rates to dabrafenib and trametinib (BRAF-mut and MEK inhibitors, respectively) and longer progression-free survival and overall survival compared with patients with detectable BRAF-mutated ctDNA. The combination of Dabrafenib with Trametinib was first approved by the FDA in 2014 to treat unresectable or metastatic melanoma in adult patients with BRAF V600 mutations. Dabrafenib and trametinib are currently approved in more than 60 countries worldwide as monotherapies and in combination for the treatment of patients with unresectable or metastatic melanoma with a BRAF V600E/K mutation. In 2018, the FDA approved the combination dabrafenib/trametinib as an adjuvant treatment for BRAF V600E-mutated, stage III melanoma after surgical resection based on the results of the COMBI-AD phase 3 study, making it the first oral chemotherapy regimen that prevents cancer relapse for node positive, BRAF-mutated melanoma.
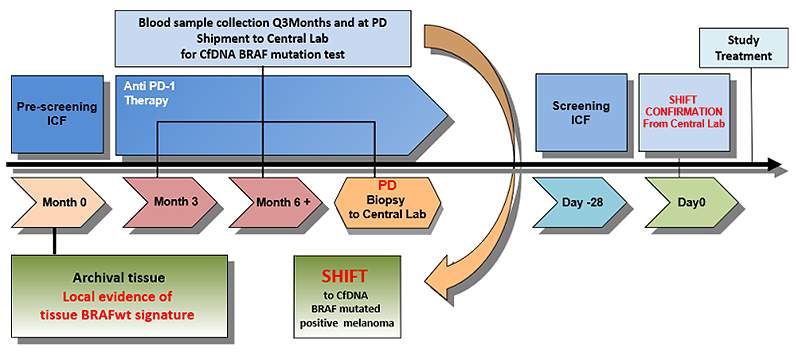
This phase II trial has been designed in order to evaluate the activity of Dabrafenib and Trametinib combination in BRAF wild type metastatic melanoma patients with a molecular shift to circulating BRAFV600 mutated free DNA upon progression to an anti PD-1 therapy.
In the clinical context of tissue BRAFwt melanoma patients, both new patients planned to receive an anti PD-1 therapy as well as those who have already started an anti PD-1 therapy will be consented and screened for this trial, in order to detect the presence of a molecular shift to BRAFV600 mutated circulating free DNA. Blood sample collection will be performed at baseline and every 3 months and circulating tumor DNA (ctDNA) will be screened for BRAF mutations using two molecular approaches:
- a real-time quantitative PCR (rtPCR) Idylla assay;
- a droplet-digital PCR (ddPCR) assay.
Tissue molecular analysis (archival tumor tissue with evidence of BRAFwt and tumor biopsy at progression) and CfDNA will be centralized to avoid result discrepancies due to different methodologies for BRAF mutation detection. In case of a mutational result discrepancy, a method of high sensitivity screening molecular analysis (Next Generation Sequencing) will be used.
Primary Objectives: The Overall Response Rate to Dabrafenib and Trametinib in BRAF wild type metastatic melanoma patients with a molecular shift to circulating BRAFV600 mutated free DNA upon progression to an anti PD-1 therapy.
Secondary Objectives are to evaluate the effects of the combination treatment on: Total Prossion Free Survival; Overall Survival; Toxicity of the investigational medicinal products (IMPs); Quality of life by means of the 30-item European Organisation for Research and Treatment of Care quality of life questionnaire (EORTC QLQ-C30).
Partners:
Dr. Paolo A. Ascierto, Istituto Nazionale dei Tumori, Fondazione “G.Pascale”, Napoli (PI); Prof. Giuseppe Palmieri IRGB- CNR- Sassari (co-PI); Mario Mandalà, University of Perugia (co-PI); Italy