Project Description
DISTINCTIVE Study – Clinical Trial
SeconD-line folfiri/aflIbercept in proSpecTIvely stratified, anti-EGFR resistaNt, metastatic coloreCTal cancer patIents with RAS Validated wild typE status
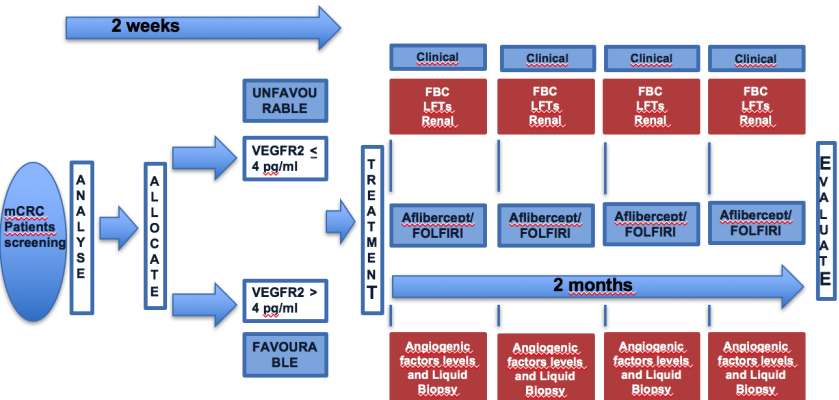
The study will be a biologically enriched, prospectively stratified phase II trial in RAS wild type metastatic colorectal cancer patients progressing after first-line treatment with oxaliplatin, fluoropyrimidines and an anti-EGFR monoclonal antibody. Eligible patients will be prospectively allocated to either of two groups according to VEGFR2 levels (ELISA-based technique, pg/ml) at study entry. Others angiogenetic factors levels concentration before and during treatment. VEGF, PlGF, HGF, VEGFR1, IL8, IL1a, T-cad, VEGFR3, SAP, VDBP, neuropilin1, CRP, endoglin plasma concentrations will be evaluated before each cycle according to an ELISA-based technique. All patients will undergo a blood test for retrieving circulating tumor DNA (Liquid Biopsy) at selected time-points before and during treatment for determining whether the status of selected tumor biomarkers evolve during tumor progression by comparing different ctDNA samples. All patients will receive aflibercept in combination with FOLFIRI according to the Italian label.
The aim of the study will be: a) to test the efficacy of aflibercept in combination with FOLFIRI (administered according to the Italian label) in the second-line treatment of RAS wild type metastatic colorectal cancer patients receiving first-line therapy with FOLFOX in combination with an anti-EGFR (either panitumumab or cetuximab); b) to prospectively validate VEGFR2 plasma levels as predictive factor for efficacy of Aflibercept in combination with FOLFIRI in the study population.
Partners:
Prof. Mario Scartozzi, Medical Oncology Unit, University of Cagliari, Cagliari (PI); Dr.Marco Puzzoni, Medical Oncology Unit, AOU, Cagliari; Prof. Giuseppe Palmieri, Unità Genetica dei Tumori CNR, Sassari; Italy